Looking for a CDx Partner in Japan?
We Can Help You Accelerate Product Launches in the Japanese Market
From development through manufacturing and sales, we can help you enter the Japanese market quickly and easily through our full service support, experience in PMDA and regulatory affairs, and extensive KOL Network.
Download our White Paper to learn more about how we can help.
About MBL
MBL offers clinical diagnosis and research reagents that measure biomarkers such as disease related proteins and/or genes related to human diseases.
Over the past 50 years, we have been engaged in-house R&D, marketing, manufacturing, sales and post-sales follow-ups. We have achieved a significant number of newly registered in vitro diagnostic products and approvals of medical reimbursement in Japan.
We are the partner of choice to provide "One Stop Shop" services for IVD/CDx products.
One Stop Shop
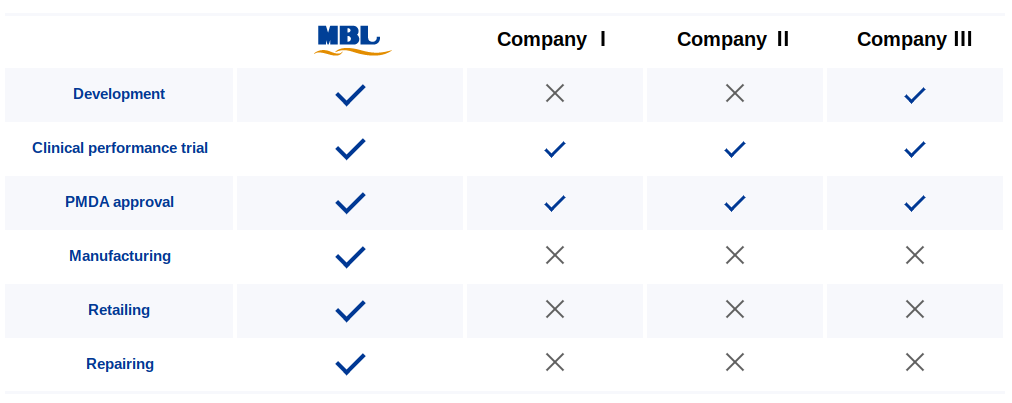
MBL has all the required business licenses for medical device manufacturing, marketing, and sales of IVD/CDx products and repairing of medical devices in Japan. We are uniquely positioned to support foreign manufacturers to entry into Japanese market.
A Leader in New IVD Medical Reimbursement Approval
MBL has an extensive, proven track record of obtaining approvals for both established and novel biomarkers. We have many years of experience in PMDA regulatory affairs and clinical developments related to IVD/CDx products.
Extensive KOL Network
MBL is one of the few IVD manufacturers participating in SCRUM-Japan, a consortium established by Japanese core hospitals and pharmaceutical companies to deliver optimal medical care to every cancer patient.
Your Partner in CDx. Expert Support.
Full Service Support
From development to retailing and repairing we've got you covered. We can take you through the process from beginning to end, or you can leverage any of our experience in specific activities. Our process is flexible and customized for your needs.
New Product Development
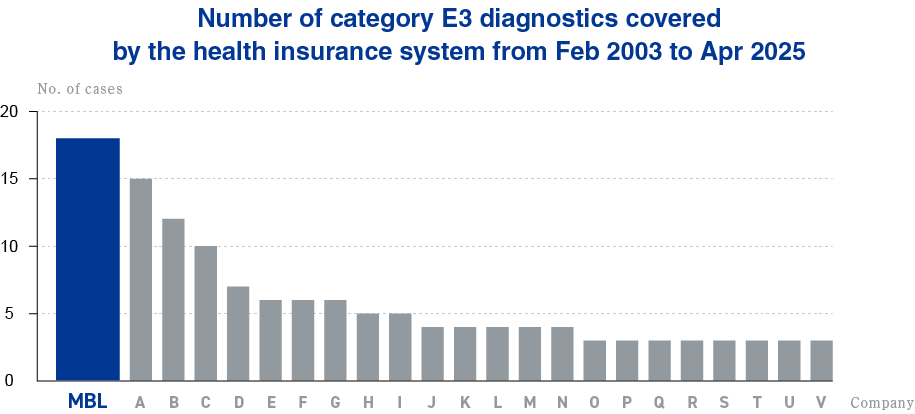
for the number of new IVDs Medical reimbursement approval
Applications for diagnostics to be covered by the health insurance system fall into three categories: E1, E2, and E3. E1 is diagnostics with previously existing measurement items and measurement methods, E2 is diagnostics with new measurement methods, and E3 is diagnostics with new measurement items. At MBL, we work with all three categories, but we are particularly strong in E3.
KOL Network Benefits
THE ONE OF THE FEW IVD COMPANIES participating in SCRUM-Japan. Industry-academia nationwide cancer genome screening project "SCRUM-Japan" is the world’s most advanced project of genetic alteration screening project for patients with cancers.
Managed by the National Cancer Center Hospital East in collaboration with medical institutions and pharmaceutical companies all over the country, the aim is to provide the best medical care to every patient.
.png)
Success Stories
MEBCDX™ AAV9 Test
Application and approval of CDx for onasemnogene abeparvovec-xioi in Japan
MEBRIGHT™ NUDT15 Kit
A expedited review was recommended because of clinical necessity
MEBGEN™ RASKET-B Kit
Quick approval of Multiplex assay
Want to learn more about how you can accelerate your product launch in Japan?
Download our white paper today!
Frequently Asked Questions
Can you develop a CDx for our medicines in Japan?
MBL can handle seamlessly - from assay system development, to application, manufacturing, and sales.
Can you develop and launch CDx concurrently with our drug approval?
MBL provides consultation for simultaneous release.
We want to measure a biomarker but don't have the measurement system for it.
Based on a wealth of know-how in developing diagnostics, MBL will construct the optimal measurement system.
We want to sell diagnostics approved overseas in Japanese market.
MBL can help with application work, manufacture and sales in the Japanese market.
LEARN MORE about entering the Japanese Market Today
From development through manufacturing and sales, we can help you enter the Japanese market quickly and easily through our full service support, experience in PMDA and regulatory affairs, and extensive KOL Network. Download our white paper to learn more about how we can help.


